A cleanroom conference in Singapore focussing on the latest trends and insights for pharma, biopharma and hitech manufacturers in Asia

5-6 December 2023
Marina Bay Sands
THANK YOU FOR JOINING US
SEE YOU IN 2024
To attend or exhibit at next year’s event, contact Brian Lubieniecki-Heyburn today! Email brianh@hpcimedia.com or call +44 (0) 207 193 8253.
If you have not received this year’s presentation slides, please email amandine@hpcimedia.com.
The two day event addresses topics ranging from standards and regulations to operational aspects of cleanrooms in pharma and semiconductor manufacturing. Attracting leading professionals from Singapore and the surrounding regions, Cleanroom Technology Conference Singapore provides the perfect place to discuss, network and source products for cleanroom and contamination control processes.
2023 EXHIBITORS

STAND B4
Interested in booking your exhibitor stand? Contact brianh@hpcimedia.com or call +44 (0) 207 193 8253 today!
2023 SPEAKERS
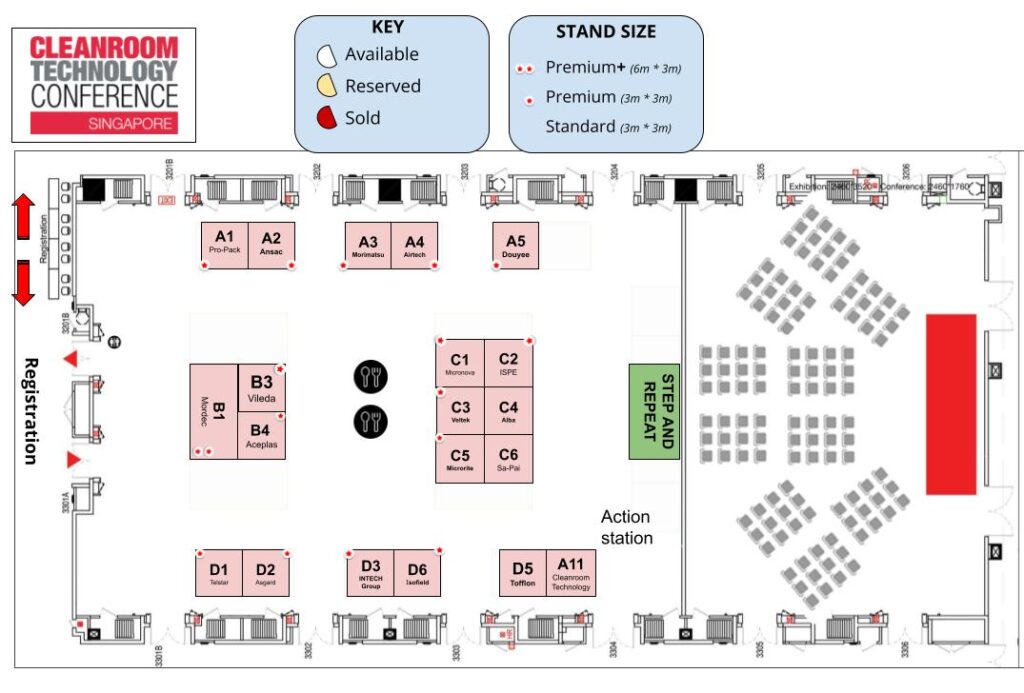
2023 FLOORPLAN
Have you heard about our UK Conference? The annual event, taking place in Birmingham, highlights the best practice and regulations affecting cleanroom design and operations across various sectors for those working within controlled environments and cleanrooms.
FAQ
No, if you have signed up and paid for your ticket it will be waiting for you at the registration desk when you arrive
Yes, we will send all the available presentations out to delegates in the weeks following the show
Yes, just email brianh@hpcimedia.com with the name of the person replacing you
Yes, all visitors to the show can attend the presentations and visit the exhibition
Yes, tea, coffee and snacks are included, as well as a hot buffet lunch.
Yes, this will be supplied to you after the show
Yes, all is included
Yes, TVs, and some furniture can be rented for the show. Speak to your Account Manager about what you need and they can provide the costs.




































